Since mRNA vaccines work by giving instructions to our cells to make the spike protein, this finding gives insight into how long the newly made protein will be present.ĭutch's team also examined the role of key host factors in cell-to-cell fusion. The study found that the majority of the spike protein degrades within 24 hours, which provides more understanding about the process of infection and vaccination. 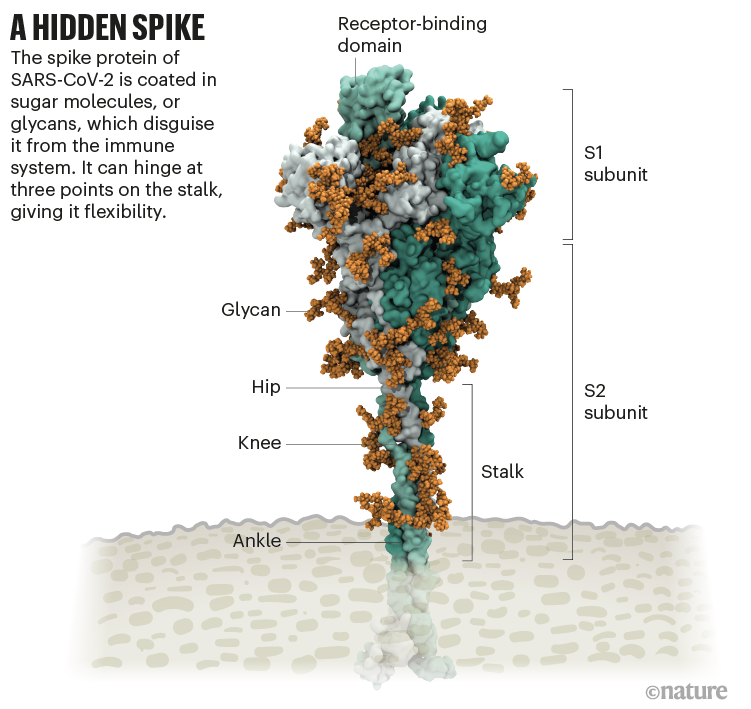
They also observed spike protein synthesis and processing in bat cells to understand if any differences were observed. 
Her team examined the effect of mutations in clinical isolates of the virus on protein stability and function. "This study gives scientists a more comprehensive understanding of how the protein works, which is significant to the continued development of vaccines and therapeutics."ĭutch's study provides insight into how stable the spike protein is, how it promotes cell-to- cell fusion and how it is modified. "The spike protein represents one of the most important therapeutic targets for COVID-19," said study lead Becky Dutch, vice dean for research in the College of Medicine and chair of the Department of Molecular and Cellular Biochemistry. Because of this function, it is the focus of most COVID-19 vaccines including the Pfizer/BioNTech and Moderna mRNA vaccines. Sources: National Center for Biotechnology Information Nature Lynda Coughlan, University of Maryland School of Medicine.The spike protein is found on the surface of SARS-CoV-2, the virus that causes COVID-19, and is responsible for its entry into host cells. March 26 India cuts back on exports of the Oxford-AstraZeneca vaccine, as infections surge in the country.Īpril 7 Britain says it will curb the use of the Oxford-AstraZeneca vaccine in adults under 30, because of the risk of rare blood clots.Īpril 9 Unusual antibodies may have caused the rare blood clots in some people who received the Oxford-AstraZeneca vaccine.Īpril 23 Researchers are examining how components of the Oxford-AstraZeneca vaccine might disrupt the normal blood clotting process under certain rare conditions.Īpril 26 The European Commission announces it has filed a lawsuit against AstraZeneca for breach of contract, for delays in shipping hundreds of millions of doses. March 22 Results from a large clinical trial show the vaccine has an overall efficacy of 79 percent. March 18 The European Medicines Agency says the Oxford-AstraZeneca vaccine is safe. March 11 Denmark, Iceland and Norway suspend the use of the vaccine because of concerns about a possible increased risk of blood clots. 3, 2021 India authorizes a version of the vaccine called Covishield, made by the Serum Institute of India. 30 Britain authorizes the vaccine for emergency use. 11 AstraZeneca announces that it will collaborate with the Russian creators of the Sputnik V vaccine, which is also made from adenoviruses.ĭec. 8 Oxford and AstraZeneca publish the first scientific paper on a Phase 3 clinical trial of a coronavirus vaccine.ĭec. 7 The Serum Institute of India announces that it has applied to the Indian government for emergency use authorization of the vaccine, known as Covishield in India.ĭec. Prime Minister Boris Johnson of Britain holds a vial of the vaccine. 12 The clinical trial resumes in the U.K. 8 The news about paused trials becomes public. Neither AstraZeneca nor Oxford announce the pause. 6 Human trials are put on hold around the world after a suspected adverse reaction in a British volunteer. 18 A Phase 3 trial of the vaccine begins in the United States, with 40,000 participants. July 30 A paper in Nature shows the vaccine appears safe in animals and seems to prevent pneumonia.Īug. June 28 A Phase 1/2 study begins in South Africa. June 23 A Phase 3 trial begins in Brazil. Some of the volunteers accidentally receive half of the intended dose. May 28 A Phase 2/3 trial of the vaccine begins in Britain. government pledges up to $1.2 billion to help fund AstraZeneca’s development and manufacturing of the vaccine. John Cairns/University of Oxford/Agence France-PresseĪpril 30 Oxford partners with AstraZeneca to develop, manufacture and distribute the vaccine. A vial of the Oxford-AstraZeneca vaccine.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
